Each year, 20m people are diagnosed with cancer. Various organs can be affected, and cancer types differ greatly at the cellular and molecular level. In about half of all cases, however, the protein p53 is mutated.
Known as the “guardian of the genome,” it plays a central tumour suppressor role: in healthy cells, it ensures that DNA damage is detected and repaired. If this is not successful, the affected cell is selectively eliminated through apoptosis – an important protection against cancer. Conversely, cells can often only develop into tumour cells when the protein p53 is inactivated by a mutation. In many cases, it becomes unstable because of the mutation and loses its functional structure.
Due to the central importance of p53, therapeutic approaches have been sought for decades to reactivate such p53 mutants—in the hope that this will cause cancer cells to die in a targeted manner. Researchers at University Medicine Frankfurt are currently testing whether p53 can be restored in ovarian cancer using introduced, intact mRNA constructs. The mRNA is packaged in lipid nanoparticles, a technology that achieved its breakthrough in the development of SARS-CoV-2 vaccines.
Other approaches aim to stabilize the mutated p53 so that it can regain its function. A first clinical success was recently reported for the substance Rezatapopt, which reactivates a specific mutation in p53 and thereby inhibits the growth of tumours with that mutation. The problem: more than 2,000 different mutations have already been described in tumours, and only a small fraction is accessible to small molecules such as Rezatapopt.
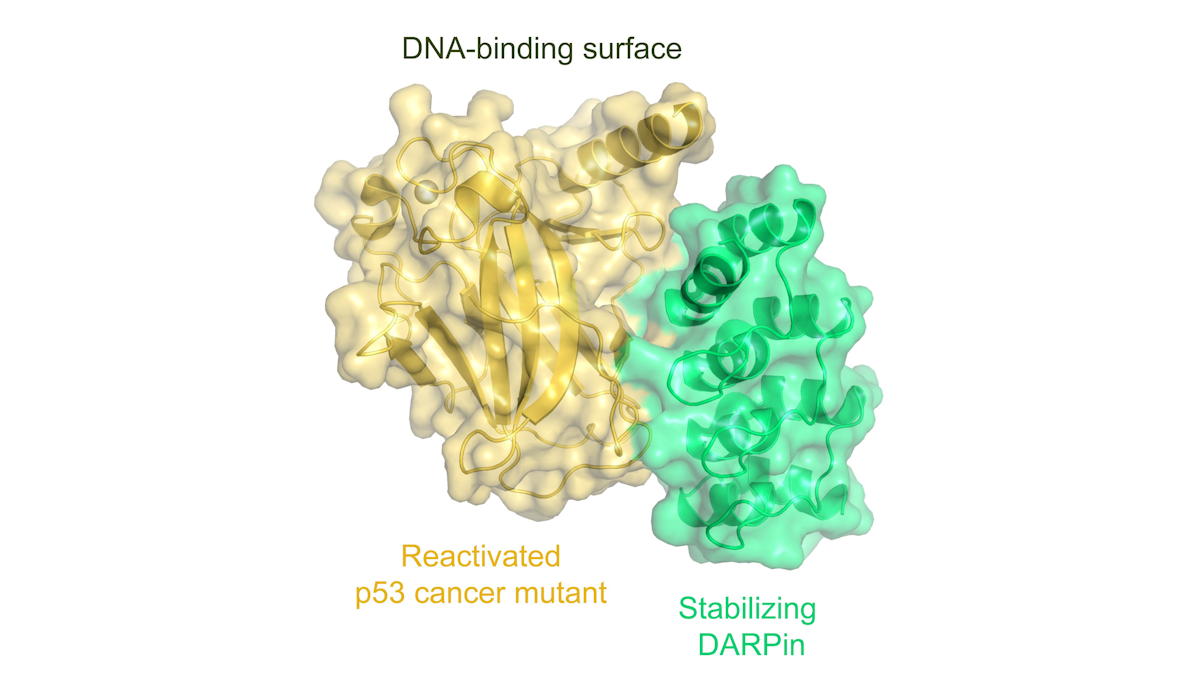
A consortium comprising research groups from Goethe University Frankfurt, Philipps University Marburg, the University of Cologne, and the University of Zurich has now developed a novel method that can reactivate many p53 mutants in the laboratory. Instead of synthesising small molecules, the consortium relies on the development of small proteins, DARPins, which bind to p53 very selectively and with high affinity, like miniature antibodies. The scientists were able to show that this binding enables many p53 mutants to regain sufficient stability, thereby restoring p53 functionality.
Volker Dötsch from the Institute of Biophysical Chemistry at Goethe University said: “The major advantage of our approach is that one of these miniature antibodies stabilizes not just a single, but many different p53 mutants, meaning it could potentially be used against various types of tumours. This also suggests that it may not be necessary to develop a specific therapeutic agent for each individual mutant.”
Antibodies have long been used successfully in cancer therapy, but until now exclusively for binding to target proteins outside the cell. The results presented here open up a new approach by offering a perspective for the use of protein-based therapeutics inside the cell as well.
The co-organizer of the study, Andreas Joerger, from the Institute of Pharmaceutical Chemistry at Goethe University, said: “In the future, our p53-reactivating DARPins could be introduced directly into tumor cells via corresponding mRNA templates packaged in lipid nanoparticles.”
The consortium partners now aim to further develop this technology for the therapeutic application of p53 stabilisers.

