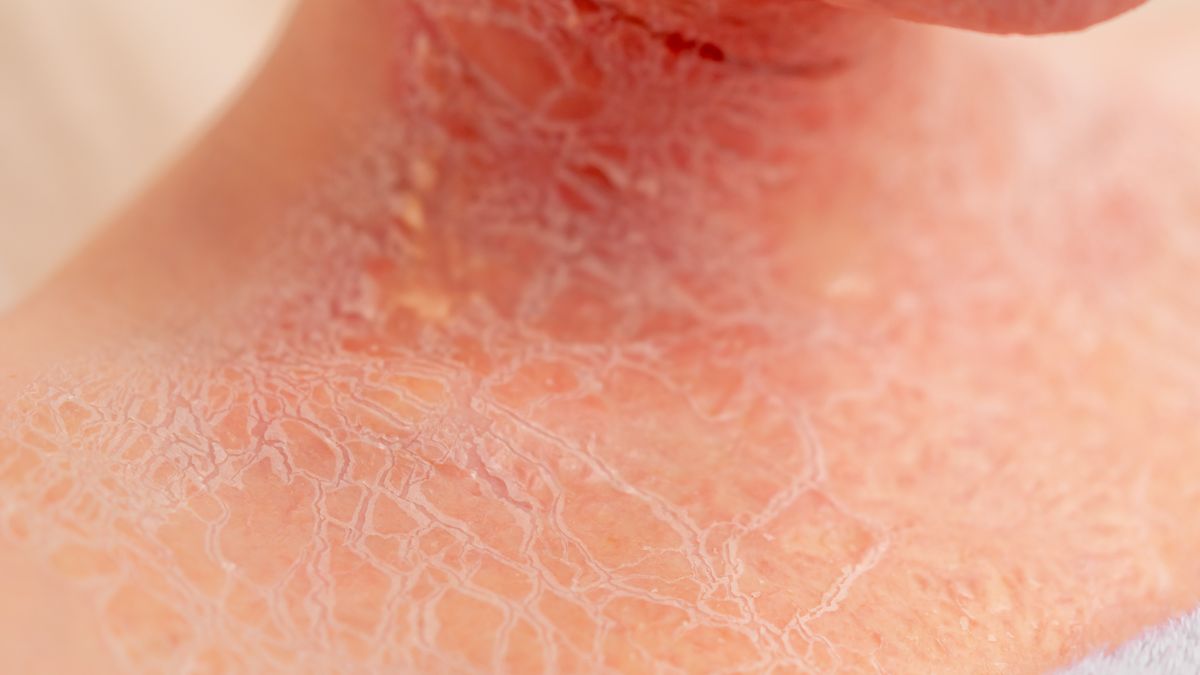
Quoin Pharmaceuticals Ltd. has announced that the U.S. Food and Drug Administration (FDA) has granted Fast Track Designation to QRX003 lotion (4%) for the treatment of Netherton syndrome, a rare and severe genetic skin disorder for which there are currently no approved treatments.
Netherton Syndrome is a rare, inherited skin disorder caused by mutations in the SPINK5 gene, leading to severe skin barrier dysfunction, chronic inflammation, and a heightened risk of infections and allergic complications. Patients often experience widespread skin redness, scaling, persistent itching, and significant impairment in quality of life. There are currently no FDA-approved therapies for the treatment of Netherton syndrome, and treatment options are limited to supportive care and off-label therapies.
“We believe that the FDA’s decision to grant Fast Track Designation to QRX003 reflects the urgent unmet need faced by patients and families living with Netherton Syndrome,” said Michael Myers, CEO and co-founder of Quoin Pharmaceuticals.
“Fast Track status enables more frequent communication with the FDA and the potential for accelerated regulatory review pathways, which may help bring the first approved treatment for Netherton Syndrome to patients as quickly as possible.”
QRX003 lotion (4%) is currently being evaluated in two late-stage whole-body clinical trials designed to assess safety and efficacy in patients with Netherton syndrome.
QRX003 previously received Orphan Drug Designation from both the U.S. FDA and the European Medicines Agency (EMA) for the treatment of Netherton Syndrome, providing potential benefits including market exclusivity upon approval, tax credits for clinical testing, and certain regulatory fee reductions.
QRX003 has also been granted Pediatric Rare Disease Designation by the FDA.