A research group led by Cecilia Sahlgren at Åbo Akademi University in Finland and the InFLAMES Research Flagship has identified a new mechanism directing the adverse remodelling of tumour tissue during breast cancer progression.
The discovery could offer new treatment opportunities against aggressive forms of breast cancer which currently lack targeted therapy options.
Breast cancer is the most common cancer among women worldwide. Localised, early-stage breast cancer has a good prognosis, but once cancer spreads to distant organs and forms metastasis, patient survival rate drops dramatically. Breast cancer subtype and the hormone receptor status of the cancer cells are major factors affecting patient survival, while the ability of cancer cells to communicate with other cells in the tumour and then in cooperation shape the surrounding tissue to their advantage is increasingly recognised as a key factor in regulating cancer metastasis and drug resistance.
One of the proteins mediating cell–cell communication called Jagged1 has been shown in previous studies to be highly expressed in aggressive, hormone receptor negative breast cancer. However, the exact functions of Jagged1 and whether it participates in breast cancer progression remained unknown.
“In our new study, we discover that Jagged1 increases breast tumour growth and metastasis and worsens survival prognosis among patients with aggressive breast cancer” said doctoral researcher Marjaana Parikainen from the Biosciences and Drug Research program at Åbo Akademi University.
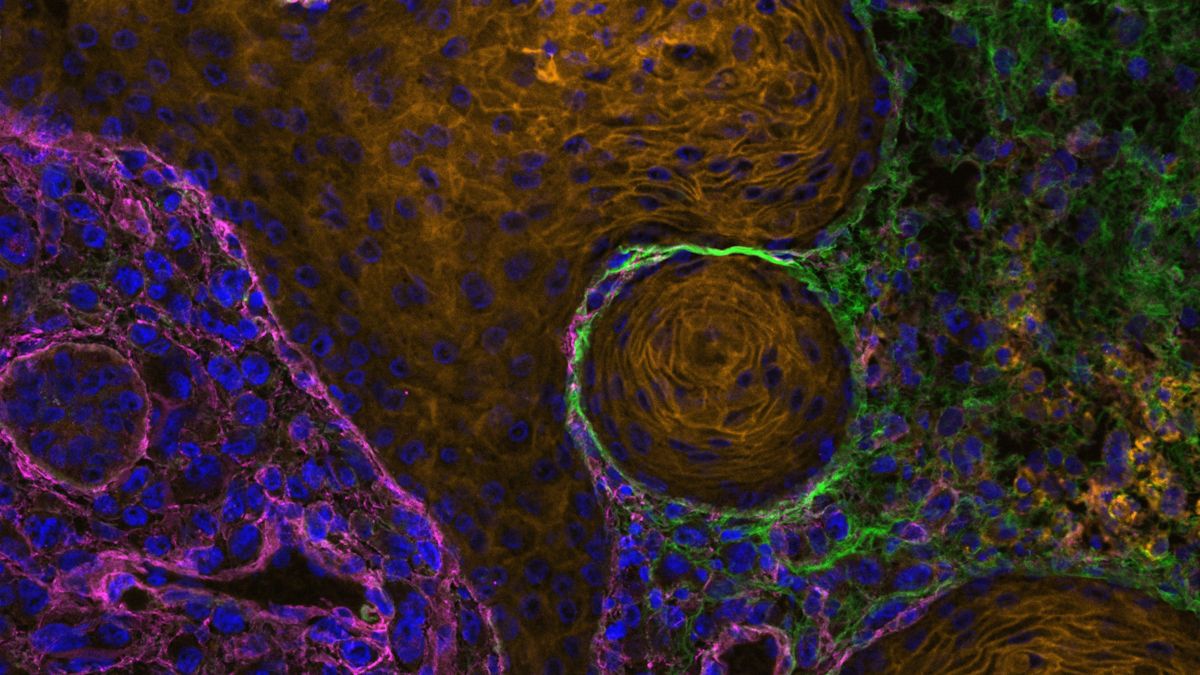
Using various cancer model systems and utilizing data from breast cancer patients, the researchers discovered a previously unknown cell communication mechanism between breast cancer cells and fibroblasts, mediated by Jagged1. Fibroblasts are a cell type responsible for producing and controlling the extracellular matrix, a structural network of molecules providing support and signalling cues for cells in tissues.
“When the cancer cells expressed Jagged1 on their cell surface, it activated the nearby fibroblasts to produce more extracellular matrix, such as collagen. In addition, these Jagged1-activated fibroblasts modify the structure of the surrounding extracellular matrix, creating highly aligned matrix fibres along which the cancer cells can move as they metastasize,” Parikainen said.
The study delves into the mechanisms by which Jagged1 regulates tumour tissue remodelling. The researchers discover that high expression of Jagged1 leads to activation of another cell–cell signalling system called the transforming growth factor beta pathway (TGFβ). TGFβ is previously known to promote tumour progression in late-stage breast cancer and is a master regulator of extracellular matrix remodelling, driving fibrosis, increased tumour stiffness, and metastasis.
“High Jagged1 promoted TGFβ activity, which in turn increased collagen deposition and matrix linearisation. In addition, we showed that cancer cells start expressing even more Jagged1 in stiffer matrix environments. Previous studies have shown that TGFβ also drives Jagged1 expression. This all points to a vicious cycle that keeps promoting itself and further tumour progression,” Parikainen said.
The study was carried out in collaboration with Jyrki Heino’s research group at the University of Turku, Finland.
The research article was published in Science Advances.

